Product Consultation
Your email address will not be published. Required fields are marked *


Content
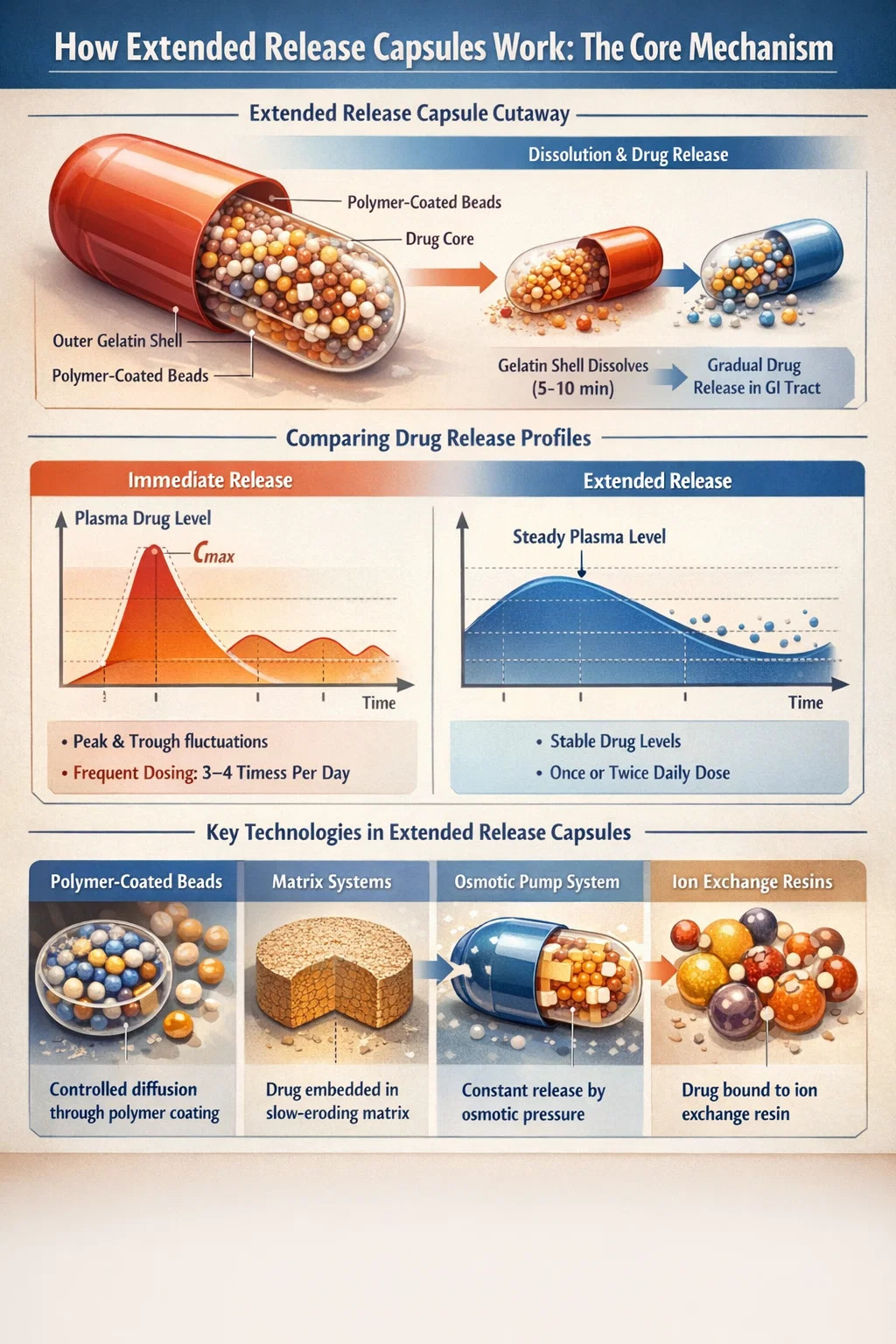
Extended release capsules are designed to release their active ingredients gradually over a defined period — typically 8 to 24 hours — rather than all at once. The result is a steadier drug concentration in the bloodstream, reduced dosing frequency, and fewer side effects caused by peak-and-trough fluctuations. This is the fundamental value proposition that makes extended release technology one of the most important advances in modern pharmaceutical manufacturing.
At the core of this mechanism is the gelatin capsule shell — a precisely engineered enclosure that controls not just containment, but the timing and location of drug release inside the gastrointestinal tract. The shell, the internal bead structure, and any polymer coatings all work in concert to ensure the drug reaches the bloodstream at the right rate, at the right time.
Unlike immediate release tablets that dissolve quickly in the stomach and deliver the full dose within 30 to 60 minutes, extended release capsules use physical and chemical barriers to slow that process. Depending on the design, a single capsule can maintain therapeutic drug levels for 12 or even 24 hours — replacing what might otherwise require three or four separate doses.
The gelatin capsule plays a far more active role in extended release delivery than most people realize. It is not simply a container — it is part of the release system itself. There are two primary types used in extended release formulations: hard gelatin capsules and soft gelatin capsules, each with distinct properties suited to different drug types and release strategies.
Hard gelatin capsules consist of two cylindrical halves — a body and a cap — that fit together to enclose a dry fill. In extended release applications, that fill is most commonly a collection of coated multiparticulate beads or mini-tablets. The gelatin shell itself dissolves quickly in gastric fluid (usually within 5 to 10 minutes), but the contents inside — each individually coated bead — continue to release the drug over many hours.
This separation of function is deliberate: the gelatin capsule shell acts as a reliable, fast-opening outer layer, while the coated internal particles carry out the extended release function. Because each bead behaves independently, this multiparticulate system also provides more consistent absorption across patients compared to a single-unit matrix tablet.
Soft gelatin capsules (softgels) are sealed, one-piece units typically filled with liquid or semi-solid formulations. In extended release designs, the gelatin shell itself can be modified — for instance, by increasing shell thickness or adding hydrophilic polymers — to slow the rate at which the shell dissolves and the drug is released. However, softgel-based extended release is less common than hard capsule approaches because controlling liquid diffusion rates is technically more challenging.
Multiple technologies can be used inside a capsule to achieve extended release. Manufacturers choose between them based on the drug's solubility, molecular weight, therapeutic window, and target release profile. The most widely used approaches are:
This is the most prevalent technology in extended release hard gelatin capsule products. Drug-loaded pellets or beads (typically 0.5–2 mm in diameter) are coated with a semi-permeable polymer film — often ethylcellulose or acrylic-based polymers like Eudragit® RS or RL. When the bead contacts gastrointestinal fluid, water slowly diffuses through the coating, dissolves the drug inside, and the drug solution then diffuses outward through the same membrane.
By varying coating thickness and polymer composition, manufacturers can engineer beads with different lag times and release rates. A single capsule may contain two or three populations of beads with different coating thicknesses — some releasing within 2 hours, others at 6 hours, and others at 12 hours — to create a smooth, sustained plasma concentration profile over the full dosing interval.
In matrix-based extended release, the drug is dispersed uniformly throughout a polymer or wax matrix. As the matrix hydrates or erodes in the GI tract, the drug slowly diffuses out. Hydrophilic matrix systems using hydroxypropyl methylcellulose (HPMC) are common; as the outer layer hydrates, it forms a gel barrier that controls how quickly drug molecules can escape to surrounding fluids.
Matrix mini-tablets can be filled into gelatin capsules alongside immediate release beads, giving formulators precise control over both the initial dose spike (to achieve rapid onset) and the sustained maintenance dose that follows. This biphasic approach is common in extended release pain medications and cardiovascular drugs.
Osmotic systems use osmotic pressure — rather than diffusion or erosion — to drive drug release at a zero-order rate (constant release regardless of drug concentration). A semi-permeable membrane surrounds a drug core and an osmotic agent. As water enters through the membrane, it generates internal pressure that pushes drug solution out through a laser-drilled orifice at a precise, constant rate.
While osmotic pump technology is most often associated with tablet formulations (like OROS®), it can be adapted into capsule formats. The technology is particularly valuable for drugs requiring very precise constant delivery over 24 hours, such as certain antihypertensives and psychiatric medications.
Some extended release capsule formulations bind the drug to ion exchange resin particles. In the GI tract, ions present in gastrointestinal fluids exchange with the drug bound to the resin, slowly liberating it. Release rate depends on ion concentration gradients along the GI tract, making this approach especially useful for liquid-filled capsules containing drug-resin complexes. Dextroamphetamine and certain antihistamines use this mechanism.
The physical and chemical properties of the gelatin capsule shell directly influence how the drug is initially exposed to GI fluids. Standard gelatin — derived from collagen hydrolysis, typically from porcine or bovine sources — dissolves rapidly in gastric conditions (pH 1–3) and at body temperature. This predictable dissolution behavior is one reason gelatin has remained the dominant capsule shell material for over a century.
However, for certain extended release applications, unmodified gelatin is modified or replaced:
The choice between gelatin and HPMC is not purely about release rate. Gelatin capsules have superior mechanical strength, lower brittleness at low humidity, and better compatibility with many moisture-sensitive fill materials. For extended release products that need to maintain structural integrity through complex manufacturing processes — such as hot-melt extrusion or fluid bed coating — the gelatin capsule's physical robustness is often decisive.
The clinical rationale for extended release capsules becomes clearest when you look at pharmacokinetic comparisons. Below is a representative comparison of an immediate release versus extended release formulation for a hypothetical drug with a 6-hour half-life:
| Parameter | Immediate Release | Extended Release Capsule |
|---|---|---|
| Time to peak concentration (Tmax) | 1–2 hours | 4–8 hours |
| Peak concentration (Cmax) | High (risk of side effects) | 30–50% lower |
| Duration of therapeutic effect | 4–6 hours | 12–24 hours |
| Doses per day | 3–4 | 1–2 |
| Peak-to-trough fluctuation | High | Low (more stable levels) |
| Patient adherence | Lower (more doses to remember) | Higher (once or twice daily) |
Research consistently shows that patient adherence improves by 20–30% when dosing frequency drops from three times daily to once daily. For chronic conditions like hypertension, epilepsy, and ADHD, this adherence improvement translates directly into better long-term outcomes and fewer hospitalizations.
Producing extended release capsules is significantly more complex than manufacturing standard immediate release forms. Each stage must be tightly controlled to ensure consistent release profiles across every batch.
Drug-loaded beads are typically produced by layering the drug solution onto sugar spheres or microcrystalline cellulose (MCC) cores using a fluid bed coater. The process, called drug layering, deposits thin, uniform layers of drug onto starter cores until the target drug loading is achieved. Bead size uniformity — typically within ±10% of the target diameter — is critical because release rate depends partly on surface area, which is directly related to particle size.
After drug loading, beads are returned to the fluid bed coater where a controlled polymer coating is applied. Coating thickness — often expressed as percentage weight gain — directly controls release rate. A 5% weight gain coating might produce 8-hour release, while a 12% weight gain with the same polymer could extend release to 16 hours. Controlling the coating process requires precise management of inlet air temperature, spray rate, product temperature, and pan speed.
Coated beads are filled into empty hard gelatin capsule shells using automated capsule filling machines. These machines must handle the beads gently to avoid coating damage — any crack or break in the polymer membrane will cause dose dumping, where a large portion of the drug releases immediately. Fill weight accuracy is also critical: a ±3% tolerance is standard for extended release products, compared to ±5% for simpler immediate release capsules.
Every batch of extended release capsules undergoes dissolution testing — a regulatory requirement that simulates drug release in vitro. The test uses USP dissolution apparatus (typically Apparatus 1 or 2) with specified media, temperature (37°C), and rotation speed. For a 12-hour extended release product, the specification might require: no more than 30% released at 1 hour, 45–65% at 4 hours, and no less than 80% at 10 hours. This three-point specification ensures neither too-rapid nor too-slow release.
Not every drug is a good candidate for extended release formulation. Several drug properties must be evaluated before committing to this approach:
Extended release capsules are used across a wide range of therapeutic categories. Some of the most clinically important applications include:
| Drug Class | Example Drugs | Benefit of Extended Release |
|---|---|---|
| CNS Stimulants (ADHD) | Amphetamine, Methylphenidate | Single morning dose covers full school/work day |
| Antihypertensives | Verapamil, Diltiazem | Smooth 24-hour BP control, no morning surge |
| Antidepressants | Venlafaxine, Bupropion | Reduced nausea, once-daily dosing |
| Opioid Analgesics | Morphine, Oxycodone | 12-hour pain control, fewer euphoric peaks |
| Antibiotics | Clarithromycin ER, Ciprofloxacin ER | Reduced GI irritation, improved tolerability |
| Antiepileptics | Carbamazepine ER, Divalproex ER | Stable drug levels, fewer breakthrough seizures |
One of the most important safety points about extended release capsules is that the entire controlled release mechanism depends on the physical integrity of the dosage form. Crushing, breaking open, or chewing an extended release capsule destroys the release-controlling barrier, causing the full dose — intended for 12 or 24 hours — to be absorbed within 30 to 60 minutes.
This phenomenon, called dose dumping, can be life-threatening for high-potency drugs. A patient who crushes a 12-hour opioid extended release capsule and swallows the contents is receiving what is effectively a 12-hour dose in a single immediate release event. This has been the cause of numerous fatal overdoses.
There are, however, exceptions. Some extended release capsule products are specifically designed so that the capsule can be opened and the beads sprinkled onto soft food (like applesauce) for patients who have difficulty swallowing. In these cases, the beads themselves carry the extended release coating — swallowing the beads whole preserves the mechanism. The prescribing information for each specific product must be consulted before any manipulation of the dosage form.
Other important considerations include:
Pharmaceutical manufacturers increasingly evaluate both gelatin and HPMC (hydroxypropyl methylcellulose) capsule shells for extended release products. Each has distinct advantages:
For most commercial extended release capsule products, the hard gelatin capsule remains the preferred shell material due to its cost-effectiveness, manufacturing compatibility, and extensive safety and regulatory track record. The decision to switch to HPMC or another alternative is typically driven by patient population requirements (vegetarian/vegan patients), drug-excipient incompatibilities, or specific stability concerns identified during formulation development.
Regulatory agencies, including the FDA, EMA, and ICH, impose stringent requirements on extended release capsule products that go well beyond those for immediate release forms. These requirements reflect the greater complexity and patient risk associated with modified release technology.
Key regulatory expectations include:
Generally, no. Opening the capsule and taking the contents — whether powder or beads — will almost always destroy the extended release mechanism, resulting in dose dumping. A few specific products are designed so that the beads can be sprinkled on food, but this must be confirmed in the prescribing information for that exact product. Never assume it is safe to open an extended release capsule without checking.
These abbreviations all refer to variations of modified release technology. XR and ER stand for extended release. SR stands for sustained release. CD stands for controlled delivery. LA (long-acting) and CR (controlled release) are also used. While the terminology varies by manufacturer and region, all of these designations indicate that the drug releases over a prolonged period rather than immediately. The specific release profile varies by product.
Not necessarily more effective in terms of total drug exposure, but they provide a more stable and often better-tolerated plasma concentration profile. For some conditions — like ADHD, chronic pain, and hypertension — the smoother drug levels translate directly into better symptom control and fewer side effects. For others, immediate release may be preferable because the physician needs precise control over drug timing or needs rapid peak concentrations.
The gelatin capsule shell dissolves within 5 to 10 minutes of ingestion, so it has minimal impact on the overall onset of drug release. The primary determinants of onset in an extended release capsule are the drug loading profile of the contents and whether an immediate release component is included. Many extended release capsule products include a small immediate release fraction (typically 20–30% of the total dose) to achieve rapid symptom relief while the extended component maintains efficacy over time.
This is most common with osmotic pump tablet systems, not capsule-based formulations. However, with some capsule-based extended release products, empty bead shells or matrix remnants may be visible in stool. This does not mean the drug was not absorbed — it simply means the carrier matrix (which is indigestible) passed through normally after the drug was released. Patients should be reassured that seeing capsule remnants in stool does not indicate treatment failure.
Your email address will not be published. Required fields are marked *
If you would like to learn more about our products, please feel free to contact us and we will do our to assist you.
No.1 Tianzhu 3rd Road, Dufu Town, Xinchang County, Zhejiang Province
86-575 8606 0065
86-159 8825 2009
+86 159 8825 2009
+1 380 215 7432
