Product Consultation
Your email address will not be published. Required fields are marked *


Content
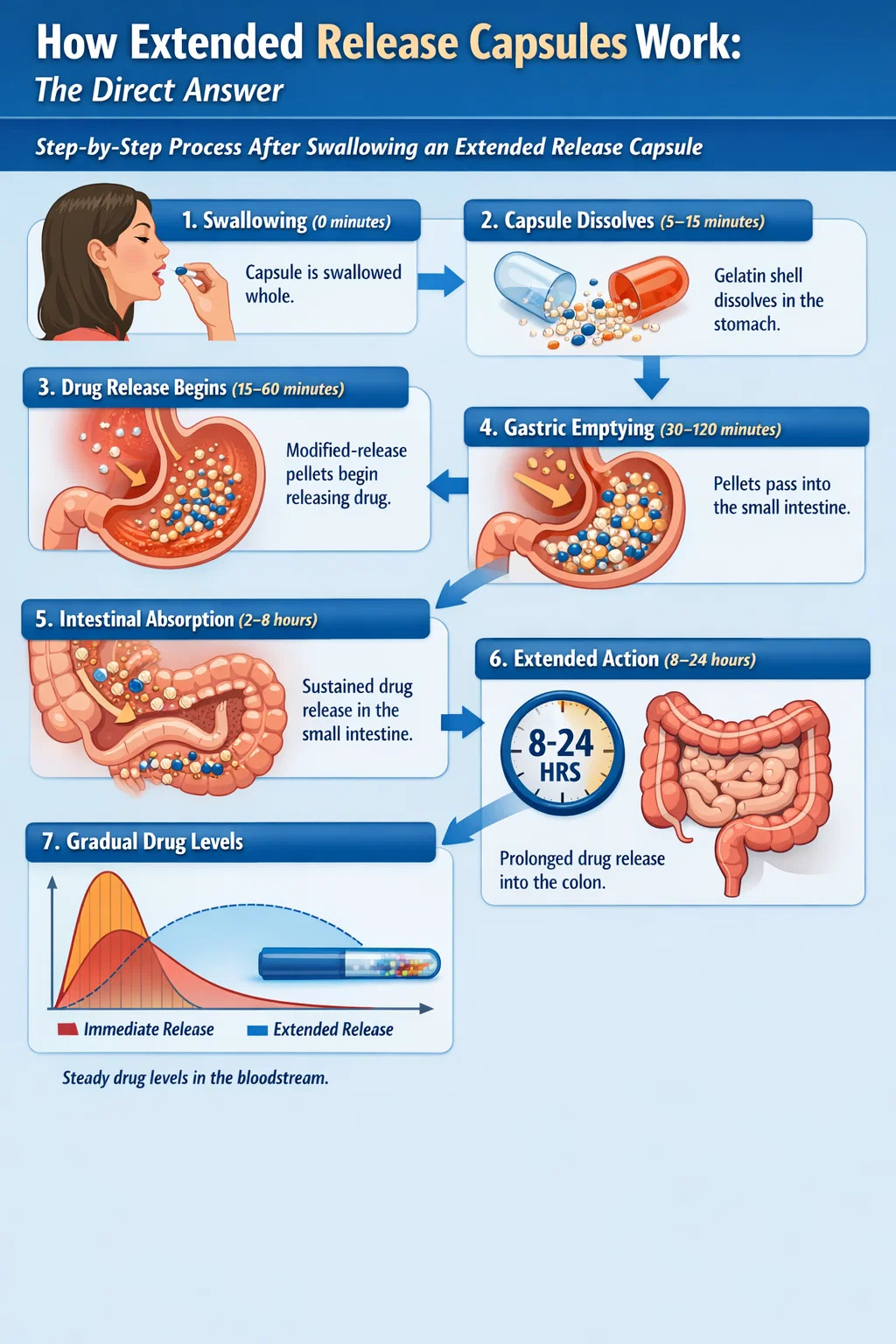
Extended release capsules work by using specialized coatings, matrices, or membrane systems inside a gelatin capsule shell to slow down the rate at which the active drug is released into the body. Instead of dissolving all at once like an immediate-release tablet, the drug is released gradually over a period typically ranging from 8 to 24 hours. This controlled delivery mechanism maintains therapeutic drug concentrations in the bloodstream for longer durations, reduces the frequency of dosing, and minimizes the peaks and troughs in drug plasma levels that often cause side effects.
The outer shell is almost always made from gelatin — either hard gelatin capsules or soft gelatin capsule variants — which dissolves quickly after ingestion, exposing the internal modified-release system to gastrointestinal fluids. The real technology lives inside, not in the shell itself.
The gelatin capsule is the first component the body encounters. Gelatin, derived primarily from animal collagen (bovine or porcine sources), forms a thin, flexible, and water-soluble shell that disintegrates within minutes after reaching the stomach. In hard gelatin capsules used for extended release formulations, the two-piece capsule (a body and a cap) encases pellets, mini-tablets, granules, or a matrix plug — all engineered to release their drug payload slowly.
Unlike immediate-release formulations where the drug itself might be compressed into a tablet exposed directly to gastric acid, the gelatin capsule shell in extended release systems serves as a protective transport vehicle. Once it dissolves — typically within 5 to 15 minutes — the internal modified-release units are liberated and begin their controlled delivery function.
Vegetarian alternatives such as hydroxypropyl methylcellulose (HPMC) capsules are increasingly used in place of animal-derived gelatin capsules, especially for consumer markets that demand plant-based or halal/kosher-compliant products. HPMC gelatin-alternative capsules behave similarly in terms of dissolution speed and have been shown in clinical comparisons to produce equivalent drug release profiles in most formulations.
Most extended release capsule products use hard gelatin capsules because they can hold solid particulate systems like coated pellets. Soft gelatin capsules (softgels) are more commonly used for liquid or semi-solid fills and are less frequently used in traditional extended release designs, though novel semi-solid matrix technologies are changing this. The distinction matters because the internal fill technology determines the release mechanism.
There is no single method for achieving extended release. Pharmaceutical scientists use several distinct mechanisms depending on the drug's chemistry, solubility, half-life, and target release duration. Understanding each mechanism helps clarify why some capsules can sustain drug levels for 12 hours while others stretch to 24 hours.
In this approach, drug-loaded pellets or granules inside the gelatin capsule are coated with a semi-permeable polymer membrane. Common polymers include ethylcellulose, Eudragit RS, and Eudragit RL. Once the gelatin capsule dissolves, gastrointestinal fluid penetrates through the membrane, dissolves the drug, and the drug solution diffuses outward through the membrane at a rate controlled by the membrane's thickness and permeability.
Membrane thickness is a primary determinant of release rate. A thicker ethylcellulose coat produces slower release; a thinner coat produces faster release. Formulators can blend fast-dissolving (Eudragit RL) and slow-dissolving (Eudragit RS) polymer grades at specific ratios to fine-tune the release profile. For example, a 70:30 RS:RL ratio might yield a 12-hour release, while a 50:50 ratio might produce 8-hour release for the same drug.
Matrix systems embed the drug within a polymer or lipid network. The drug must diffuse through the matrix material itself rather than through a surface membrane. Hydrophilic matrix systems use swellable polymers like hydroxypropyl methylcellulose (HPMC) — when the gelatin capsule dissolves and the matrix contacts gastrointestinal fluid, the HPMC swells to form a gel layer. Drug molecules must diffuse through this gel, which slows release significantly.
Hydrophobic matrix systems use inert waxes (carnauba wax, beeswax) or polymers (ethylcellulose) that do not swell but create a tortuous diffusion path. These systems are particularly useful for water-soluble drugs that would otherwise release too rapidly.
Osmotic systems use osmotic pressure as the driving force for drug release. The most widely known system is the OROS (Oral Osmotic) system developed by ALZA Corporation, now used in drugs like Concerta (methylphenidate) and Procardia XL (nifedipine). Inside the gelatin capsule shell or tablet coating is a semi-permeable membrane with a small laser-drilled orifice. Water enters through the membrane driven by osmotic pressure, which pushes the drug solution out through the orifice at a near-zero-order rate — meaning an almost constant amount of drug is delivered per unit time regardless of gastrointestinal conditions.
Osmotic systems are exceptionally resistant to food effects and pH changes, making them among the most reliable extended release technologies in clinical use. Drug release rates from osmotic capsule systems typically vary by less than 10% across gastric and intestinal pH conditions.
Some extended release capsules contain drug-resin complexes where the drug is bound to an ion-exchange resin. As the complex travels through the gastrointestinal tract, ions naturally present in digestive fluids (sodium, chloride) displace the drug from the resin, releasing it gradually. This mechanism is especially useful for liquid formulations of extended release drugs such as certain cough suppressant and antihistamine products.
Many modern extended release capsules use a multiparticulate approach — the gelatin capsule contains hundreds or thousands of tiny coated pellets (also called beads or spheroids), each between 0.5 and 2 mm in diameter. This approach has several significant advantages over single-unit systems like matrix tablets.
Drugs like diltiazem (Cardizem CD), omeprazole (Prilosec), and dextroamphetamine (Adderall XR) rely on multiparticulate systems encased in hard gelatin capsules. Adderall XR, for instance, uses a 50:50 blend of immediate-release and delayed-release beads in a single hard gelatin capsule to provide a biphasic release profile that mimics twice-daily dosing.
Not all delayed or extended release capsules release drug throughout the entire gastrointestinal tract. Enteric-coated systems use polymers that remain intact in the acidic stomach environment (pH 1–3) but dissolve rapidly when they reach the higher pH of the small intestine (pH 5.5–7.4). Common enteric polymers include cellulose acetate phthalate (CAP), polyvinyl acetate phthalate (PVAP), and methacrylic acid copolymers (Eudragit L and S grades).
In an enteric extended release gelatin capsule, the outer hard gelatin capsule shell dissolves in the stomach, but the internal enteric-coated pellets or tablets resist dissolution until they exit into the small intestine. This is clinically valuable for:
Eudragit S100 dissolves at pH 7.0 and above, making it useful for targeting the terminal ileum and colon. Eudragit L100 dissolves at pH 6.0, targeting the proximal small intestine. By blending or layering these polymers on pellets inside a gelatin capsule, pharmacists can engineer sophisticated site-specific delivery systems.
The table below summarizes the primary extended release mechanisms used in gelatin capsule-based products, their key polymers, typical release durations, and example drugs:
| Mechanism | Key Polymers / Materials | Typical Duration | Example Drug Products |
|---|---|---|---|
| Membrane Diffusion | Ethylcellulose, Eudragit RS/RL | 8–16 hours | Diltiazem CD, Verapamil SR |
| Hydrophilic Matrix | HPMC, Carbopol | 12–24 hours | Metformin ER, Oxycodone ER |
| Osmotic Pump (OROS) | Cellulose acetate | Up to 24 hours | Concerta, Nifedipine XL |
| Multiparticulate (Beads) | Eudragit, Ethylcellulose | 8–24 hours | Adderall XR, Morphine SR |
| Enteric / pH-Dependent | Eudragit L/S, CAP | Site-targeted release | Omeprazole, Delayed-release aspirin |
| Ion-Exchange Resin | Amberlite IRP69/88 | 8–12 hours | Dextromethorphan ER liquids |
Walking through the physiological journey of an extended release gelatin capsule makes the mechanism easier to visualize:
The clinical rationale for extended release capsule formulations goes beyond patient convenience. The pharmacokinetic profile produced by these systems has direct therapeutic consequences.
Immediate-release formulations produce a sharp spike in plasma drug concentration shortly after dosing — often associated with side effects. Extended release systems flatten this peak. For example, nifedipine immediate-release causes reflex tachycardia and headache due to its rapid peak concentration, while nifedipine extended release (Procardia XL) produces a smooth concentration curve that largely eliminates these side effects. The C-max of the extended release version is approximately 30–40% lower than the immediate-release version at equivalent total doses.
Extended release formulations are engineered so drug plasma levels remain above the minimum effective concentration throughout the dosing interval. For antibiotics with concentration-independent (time-dependent) killing, staying above the MEC for a greater percentage of the dosing interval directly correlates with clinical efficacy. For chronic condition management — hypertension, diabetes, epilepsy — maintaining consistent drug levels prevents therapeutic gaps that could lead to symptom breakthrough.
Reducing dosing frequency from three or four times daily to once or twice daily has a measurable impact on patient adherence. Meta-analyses of adherence data consistently show that once-daily dosing improves adherence rates by 15–25% compared to multiple daily dosing in chronic disease management. This is not a trivial difference — in diseases like hypertension, where long-term consistent control is essential to prevent stroke and cardiac events, adherence improvements translate directly into better outcomes.
Conditions like hypertension, epilepsy, asthma, and pain follow circadian patterns. Immediate-release drugs taken at bedtime may wear off during early morning hours — precisely when the risk of cardiac events or seizure activity is elevated. Extended release capsule formulations can provide 24-hour coverage from a single evening dose, addressing this clinical vulnerability.
Extended release systems are engineered under specific assumptions. Several physiological and behavioral factors can disrupt the intended release profile.
Food delays gastric emptying, which can prolong the gastric residence time of extended release systems. For most multiparticulate gelatin capsule formulations, this effect is minimal. However, high-fat meals can increase the absorption of some drugs from matrix systems — a phenomenon called a food effect. Extended release nifedipine tablets taken with grapefruit juice can see bioavailability increase by up to 34% due to CYP3A4 inhibition in the intestinal wall, an example of a pharmacokinetic food-drug interaction superimposed on an extended release system.
Conditions that accelerate gastrointestinal transit — such as Crohn's disease, short bowel syndrome, or severe diarrhea — can cause extended release pellets to exit the absorptive small intestine before releasing their full drug load, resulting in underdosing. Conversely, slowed motility (as seen with diabetic gastroparesis or opioid use) can extend residence time and increase total drug absorption beyond expected levels.
Some patients and caregivers open gelatin capsule shells to mix contents with food or liquid for easier swallowing. This practice is safe only for multiparticulate systems where the individual pellets are left intact. The entire dose can be sprinkled on soft food like applesauce without disrupting the release mechanism, as long as the pellets are not chewed. However, capsules containing matrix plugs, osmotic units, or single-unit modified-release systems should never be opened, crushed, or chewed — doing so destroys the extended release mechanism and delivers the entire dose immediately, risking dose dumping and toxicity.
Gelatin capsules are hygroscopic — they absorb moisture from the environment. Excessive humidity can cause the gelatin capsule shell to soften, stick, or deform, potentially affecting how the internal system releases drug. Most extended release capsule products should be stored below 30°C in low-humidity conditions and kept in their original packaging until use.
Drug manufacturers use a variety of suffixes to indicate modified release formulations, which can cause confusion. Regulatorily, the FDA classifies modified release products as either extended release (ER) or delayed release (DR). All the commercial suffixes below refer to variations of these two categories:
From a regulatory standpoint, the FDA requires that extended release drug products demonstrate specific in vitro dissolution profiles and in vivo pharmacokinetic data to qualify for ER designation, regardless of what marketing suffix the manufacturer chooses.
Extended release formulations come in both capsule and tablet forms. The choice between them affects patient experience, manufacturing, and sometimes clinical performance.
| Feature | Extended Release Gelatin Capsule | Extended Release Tablet |
|---|---|---|
| Shell material | Gelatin or HPMC capsule | Polymer coating over compressed tablet |
| Internal system | Pellets, granules, or matrix plug | Monolithic matrix or coated core |
| Can be opened and sprinkled | Often yes (multiparticulate) | Generally no |
| Risk of dose dumping if damaged | Lower (multiparticulate systems) | Higher (single-unit matrix) |
| Drug load capacity | Moderate (limited by capsule size) | Higher (compression allows dense fill) |
| Patient swallowing ease | Generally easier (smooth surface) | Variable (coated tablets also smooth) |
The field continues to evolve. Several emerging approaches are expanding what extended release capsule formulations can achieve.
Additive manufacturing (3D printing) enables creation of internal drug structures with complex geometries that are impossible to achieve by conventional granulation or coating. These printed structures can be encased in a gelatin capsule to produce highly precise, programmable release profiles. The FDA approved the first 3D-printed drug product (Spritam, levetiracetam) in 2015, signaling regulatory acceptance of this manufacturing approach.
For inflammatory bowel disease treatment, researchers have developed capsule systems that combine pH-dependent and time-dependent release to specifically target the colon. Systems like CODES (Colon-targeted Delivery System) use a lactulose-containing core that is fermented by colonic bacteria to trigger release — an entirely microbially-driven extended release mechanism unique to the colonic environment.
In response to the opioid epidemic, regulatory agencies now encourage or require abuse-deterrent formulations for opioid extended release products. Technologies include embedding gelling agents that render the drug non-injectable when dissolved, incorporating opioid antagonists (naltrexone) in inner layers that release only when the product is tampered with, and using physical barriers that resist crushing or extraction. These systems still rely on the gelatin capsule shell as the outer delivery vehicle, but the internal architecture is engineered with additional abuse-deterrent layers.
Chronotherapy aligns drug delivery with the body's circadian biological rhythms. Some conditions — rheumatoid arthritis, asthma, angina — peak in symptoms during the early morning hours (4–8 AM). Chronotherapeutic extended release capsule systems are designed with a programmed lag time so that a drug taken at bedtime releases its therapeutic payload precisely during these high-risk morning hours, rather than at the time of administration.
Understanding how extended release capsules function helps patients use them correctly and avoid errors that compromise their effectiveness.
Your email address will not be published. Required fields are marked *
If you would like to learn more about our products, please feel free to contact us and we will do our to assist you.
No.1 Tianzhu 3rd Road, Dufu Town, Xinchang County, Zhejiang Province
86-575 8606 0065
86-159 8825 2009
+86 159 8825 2009
+1 380 215 7432
