Product Consultation
Your email address will not be published. Required fields are marked *


Content
HPMC capsules — short for hydroxypropyl methylcellulose capsules — differ from conventional gelatin capsules in their raw material origin, physical behavior under varying environmental conditions, compatibility with sensitive active ingredients, and suitability for specific consumer markets. While gelatin capsules have dominated the pharmaceutical and supplement industry for well over a century, HPMC capsules have steadily carved out a distinct and growing position, particularly where plant-based formulation, moisture sensitivity, and broad religious or dietary acceptance are priorities.
The differences are not superficial. They extend from molecular structure to filling-line behavior to shelf-life performance. For formulators, brand owners, and consumers who need more than a standard gelatin shell, understanding these distinctions is genuinely useful — not just theoretical.
Hydroxypropyl methylcellulose is a semi-synthetic polymer derived from cellulose, the structural component found in plant cell walls. The manufacturing process involves treating purified cellulose — typically sourced from wood pulp or cotton linters — with propylene oxide and methyl chloride under controlled alkaline conditions. The result is a water-soluble, non-ionic polymer that forms a clear, flexible film when cast into capsule shells.
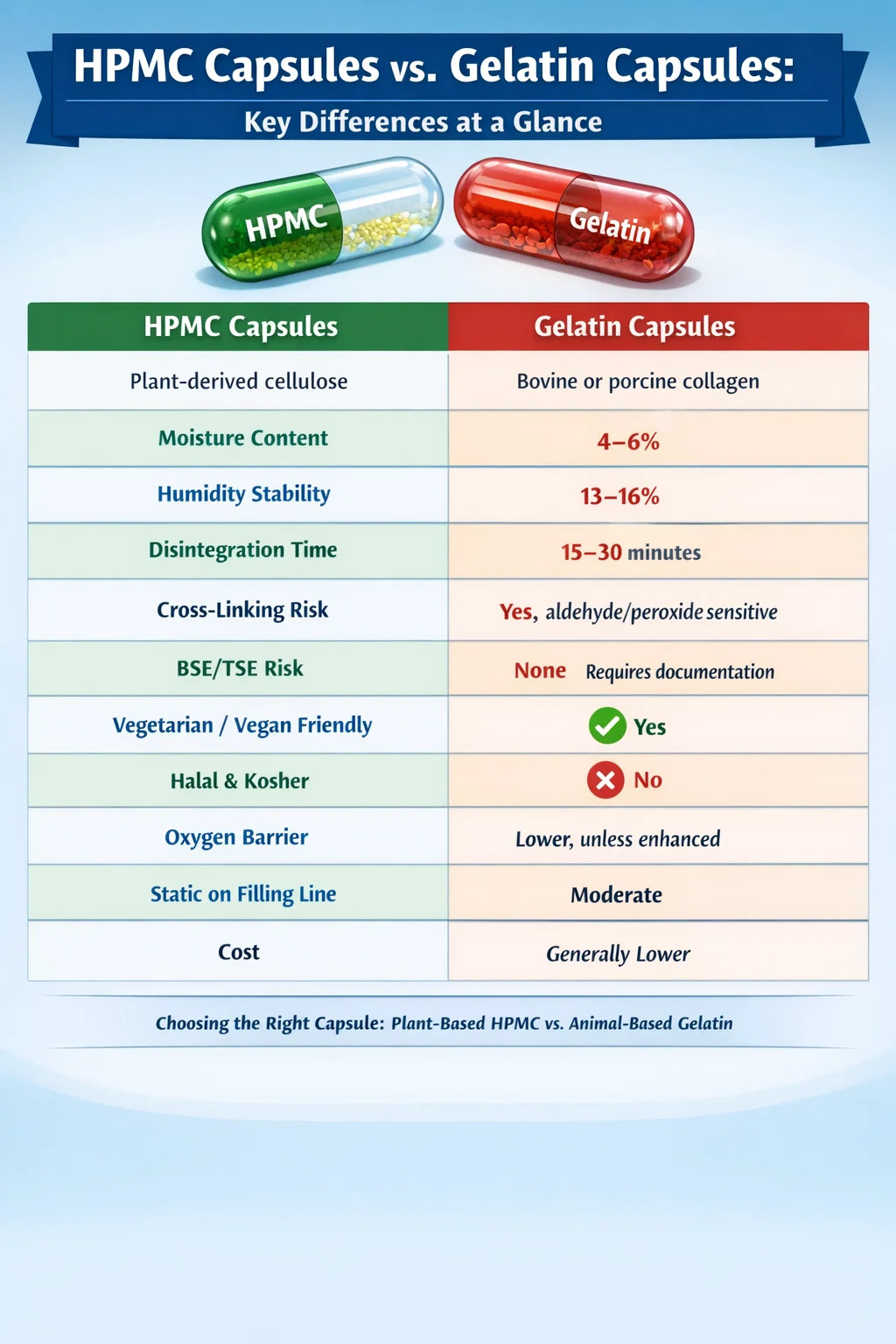
Gelatin, by contrast, is a protein derived from collagen, obtained by boiling animal connective tissue — most commonly bovine (cattle) hides and bones, or porcine (pig) skin. This animal-derived origin creates immediate limitations for vegetarian, vegan, halal, kosher, and certain religious dietary markets. HPMC is 100% plant-derived and carries no animal-origin concerns whatsoever.
The HPMC used in capsule manufacturing typically falls within specific substitution ranges. Methoxy content generally sits between 19% and 30%, while hydroxypropoxy content ranges from 4% to 12%. These parameters affect gel formation temperature, viscosity, and film strength — all of which influence how the finished capsule behaves on a filling machine and in a patient's gastrointestinal tract.
One of the most practically significant differences between HPMC and gelatin capsules is how each material interacts with moisture — both during manufacturing and over the shelf life of the finished product.
Standard gelatin capsules typically contain between 13% and 16% moisture by weight. This moisture is not incidental — it is structurally necessary to maintain the flexibility and integrity of the gelatin shell. If the surrounding humidity drops too low, gelatin capsules become brittle and crack. If humidity climbs too high, they soften, deform, or stick together. Gelatin capsules require storage at 35–65% relative humidity (RH) and are sensitive to temperatures above 40°C.
This moisture dependency creates challenges when the capsule fill contains hygroscopic (moisture-absorbing) ingredients. Substances like anhydrous magnesium chloride, certain herbal extracts, and amino acid powders can pull moisture out of the gelatin shell, causing it to become brittle and fail. Cross-linking between gelatin molecules triggered by aldehydes — including trace aldehydes found in certain excipients or fill materials — can also lead to dissolution failures even when visual appearance seems normal.
HPMC capsules contain significantly less moisture — typically between 4% and 6% by weight, depending on the manufacturer and grade. More importantly, HPMC does not require moisture for mechanical stability in the same way gelatin does. The polymer chain maintains its structural integrity across a much wider humidity range.
HPMC capsules can remain stable at relative humidity levels as low as 10% RH without becoming brittle — a performance characteristic that is practically impossible for standard gelatin shells to match. This makes HPMC capsules substantially more appropriate for:
For formulation scientists working with moisture-sensitive active pharmaceutical ingredients (APIs), this physical characteristic alone can determine whether HPMC or gelatin is the viable choice.
Dissolution — the rate and manner in which a capsule shell breaks down and releases its contents — is a critical performance parameter for any oral dosage form. HPMC and gelatin capsules dissolve through fundamentally different mechanisms.
Gelatin dissolves through a relatively straightforward hydration and enzymatic digestion process. In the stomach, gastric enzymes including pepsin begin breaking down the protein chains, while water absorption causes the shell to soften and eventually rupture. Under standard USP dissolution test conditions at 37°C in simulated gastric fluid, intact gelatin capsules typically disintegrate within 5 to 10 minutes.
However, this performance assumes the gelatin has not been cross-linked. Gelatin cross-linking — which can occur as a result of exposure to aldehydes (including formaldehyde vapor, malonaldehyde from rancid oils, or peroxides from excipients) — creates covalent bonds between protein chains that resist enzymatic digestion. The capsule may appear visually intact and normal but fail to dissolve on time or at all. Regulatory agencies including the FDA have acknowledged this phenomenon, which led to the development of two-tier dissolution testing for gelatin capsules.
HPMC capsules dissolve through a purely physical process — hydration and erosion of the polymer matrix. Because HPMC is not a protein, it has no cross-linking vulnerability from aldehydes or peroxides. The polymer swells as water penetrates the shell, and the capsule opens mechanically rather than through enzymatic digestion.
HPMC capsule disintegration typically takes 15 to 30 minutes under standard conditions — noticeably slower than gelatin under identical test conditions. For most immediate-release formulations, this difference is clinically insignificant. The API is released well within the absorption window. However, for drugs or supplements requiring very rapid dissolution onset, this timing difference should be factored into formulation development.
Some HPMC capsule manufacturers use gelling agents such as carrageenan or guar gum to improve the consistency of the capsule-forming process. These agents can affect the dissolution profile and should be declared clearly in formulation documentation, as certain regulatory bodies and consumer segments have concerns about carrageenan specifically.
A notable variant — HPMC capsules formulated for enteric release — can be engineered to resist dissolution in acidic gastric conditions and release their contents only upon reaching the higher pH environment of the small intestine. This is a significant advantage for APIs that are acid-labile or that cause gastric irritation when released in the stomach.
Capsule-fill compatibility is a formulation concern that does not always receive adequate attention until a product fails stability testing. Both gelatin and HPMC capsules have compatibility limitations, but they differ in important ways.
HPMC capsules are generally preferred or required for the following fill types:
Gelatin capsules retain advantages for certain liquid-fill applications. Because gelatin forms a tighter, more impermeable seal in some liquid-fill capsule systems, they have a longer validated track record with certain lipid-based drug delivery systems (LBDDS). HPMC liquid-fill capsule technology has advanced significantly but requires careful attention to band-sealing and fill viscosity parameters.
Oxygen transmission through capsule shells is a factor that is frequently overlooked in early formulation development but becomes important when working with oxidation-sensitive APIs — including omega-3 fatty acids, CoQ10, astaxanthin, carotenoids, and many botanical extracts.
Gelatin capsules have lower oxygen permeability than standard HPMC capsules. The dense, cross-linked protein network of a gelatin shell provides a modest but measurable barrier to oxygen ingress. Standard HPMC shells, being more hydrophilic and porous at the molecular level, allow somewhat higher oxygen transmission.
For oxidation-sensitive fills in HPMC capsules, formulators can address this by:
Some specialty HPMC capsule grades from manufacturers such as Lonza (Vcaps Plus), ACG, and Qualicaps have been engineered with enhanced barrier properties, narrowing the oxygen permeability gap with gelatin considerably.
For products targeting global markets, HPMC capsules offer meaningful regulatory and commercial advantages over gelatin capsules in several areas.
The global vegetarian and vegan supplement and pharmaceutical market has grown substantially over the past decade. Market research from multiple sources indicates that vegetarian capsule claims can increase consumer willingness to purchase, particularly in European, North American, and South Asian markets. HPMC capsules are certified vegetarian by organizations including the Vegetarian Society and are accepted across vegan labeling schemes globally. Gelatin, being animal-derived, categorically cannot carry these claims.
Halal compliance for gelatin capsules is complex. Bovine gelatin sourced from animals slaughtered according to halal requirements can qualify for halal certification, but supply chain verification is demanding and consumer trust remains variable. Porcine gelatin is categorically prohibited under Islamic dietary law. HPMC capsules achieve halal certification far more straightforwardly because the source material is entirely plant-derived and free of any animal slaughter process.
For kosher certification, similar considerations apply. Fish gelatin can be certified kosher under some schemes, but HPMC removes the complexity entirely. For products targeting markets in the Middle East, Southeast Asia, and Jewish consumer communities in North America and Europe, HPMC capsules significantly simplify certification and labeling.
Bovine Spongiform Encephalopathy (BSE) and Transmissible Spongiform Encephalopathies (TSE) represent a regulatory and reputational risk associated with bovine-derived materials including gelatin. Although current gelatin manufacturing processes — including acid and alkaline hydrolysis at high temperatures — are considered effective in eliminating BSE/TSE infectivity according to regulatory guidance, HPMC capsules carry zero BSE/TSE risk by definition because no animal-derived source material is involved at any step in their manufacture.
For pharmaceutical manufacturers supplying to regulatory authorities that require BSE/TSE risk assessments — including EMA-regulated markets in Europe — HPMC capsules can simplify the regulatory submission process by eliminating this category of risk documentation entirely.
From a manufacturing perspective, HPMC and gelatin capsules share similar external dimensions and can generally be filled on the same capsule-filling equipment, whether dosator, tamping pin, or vacuum-drum types. However, their behavior on the filling line differs in ways that production teams need to account for.
HPMC capsules generate significantly more static charge than gelatin capsules during high-speed filling operations. This is a direct consequence of HPMC's lower moisture content — water is a natural conductor that dissipates static charge. At low humidity, HPMC capsule shells can accumulate electrostatic charges that cause them to cling to machine parts, each other, and equipment surfaces, reducing filling efficiency and increasing jam rates.
Controlling filling-room relative humidity to between 40% and 55% RH and using ionizing air bars near critical filling points are standard mitigation strategies. Some HPMC capsule manufacturers incorporate antistatic agents into their shell formulations to reduce this tendency.
While HPMC capsules tolerate a wider humidity range than gelatin, they are not completely immune to brittleness at extremely low humidity (below approximately 15–20% RH). Filling operations in very dry environments or involving products filled with highly desiccating materials should monitor and manage shell brittleness proactively, though the threshold for concern is considerably lower than for gelatin.
HPMC capsules have slightly different mechanical properties from gelatin capsules when it comes to the force required to lock the cap onto the body. HPMC is somewhat harder and less elastic than gelatin. Machine operators switching between shell types typically need to adjust locking station parameters to avoid cracking or incomplete locking. This is a minor operational consideration but one worth noting for facilities that run both shell types.
The table below summarizes the key differences across the most relevant performance, regulatory, and manufacturing parameters.
| Parameter | HPMC Capsules | Gelatin Capsules |
|---|---|---|
| Source material | Plant-derived cellulose | Bovine or porcine collagen |
| Moisture content | 4–6% | 13–16% |
| Humidity stability range | 10–70% RH | 35–65% RH |
| Disintegration time (standard) | 15–30 minutes | 5–10 minutes |
| Cross-linking risk | None | Yes — aldehyde/peroxide sensitive |
| BSE/TSE risk | None | Low but requires documentation |
| Vegetarian/vegan suitability | Yes | No |
| Halal/kosher suitability | Straightforward certification | Complex, source-dependent |
| Oxygen barrier | Lower (standard); improved grades available | Moderate |
| Static generation on filling line | Higher | Lower |
| Cost | Generally higher | Lower |
HPMC capsules cost more than standard gelatin capsules. The price premium varies by grade, size, and order volume, but HPMC capsules typically cost 30% to 100% more per unit than comparable gelatin shells when purchasing at similar volumes. For high-volume commodity supplement products where margins are tight, this difference is material to business decisions.
However, the cost differential should be evaluated against the full context:
For brands operating in the natural, functional food, or premium nutraceutical space, "vegetarian capsule" as a front-panel claim has demonstrable consumer value. Consumer surveys across multiple studies consistently show that vegetarian and clean-label claims affect purchase intent among health-conscious consumers, and HPMC capsules enable those claims without compromise.
The HPMC capsule category is not monolithic. Several specialized formats have been developed to address specific formulation challenges that neither standard HPMC nor standard gelatin capsules can adequately handle.
Enteric HPMC capsules are designed to remain intact in the stomach's acidic environment (pH 1–3) and dissolve only upon reaching the small intestine (pH 5.5 and above). This is achieved either through coating the capsule shell with enteric polymers such as HPMC phthalate or cellulose acetate phthalate, or by formulating the capsule wall itself with enteric-grade materials. Applications include acid-sensitive enzymes like serrapeptase, probiotics that are vulnerable to gastric acid, and NSAIDs where gastric irritation is a known adverse effect.
Modified-release HPMC capsule systems have been developed for delivery of therapeutics to specific regions of the gastrointestinal tract, including the colon. These systems exploit pH-dependent or time-dependent release mechanisms and are relevant for inflammatory bowel disease treatments, colon cancer chemotherapy adjuncts, and microbiome-targeted formulations.
Liquid-fill technology using HPMC shells allows the encapsulation of oils, lipid-based systems, and semi-solid matrices in vegetarian shells. This format is suitable for bioavailability-enhanced lipid drug delivery systems and for nutrients such as vitamin D3, vitamin K2, astaxanthin, and omega-3 concentrates. The HPMC shell must be band-sealed after filling to prevent leakage, as HPMC does not self-seal in the way that some softgel gelatin systems do under heat and pressure.
Based on the technical, regulatory, and commercial factors reviewed above, HPMC capsules are clearly the better choice — or the only viable choice — in the following scenarios:
Gelatin capsules remain appropriate for cost-sensitive, high-volume applications with conventional fills where no dietary, religious, or compatibility constraints apply, and where rapid dissolution onset is a formulation priority. The two technologies are not in a simple hierarchy — they occupy different niches with genuine and legitimate differences in performance.
For formulation teams approaching a new product, the capsule shell selection decision should be made early — ideally during preformulation — because switching between HPMC and gelatin after stability work has begun adds cost, time, and potential retesting obligations. The physicochemical profile of the fill, the target market, the storage and distribution environment, and the regulatory filing jurisdiction should all inform that decision from the outset.
Your email address will not be published. Required fields are marked *
If you would like to learn more about our products, please feel free to contact us and we will do our to assist you.
No.1 Tianzhu 3rd Road, Dufu Town, Xinchang County, Zhejiang Province
86-575 8606 0065
86-159 8825 2009
+86 159 8825 2009
+1 380 215 7432
