Product Consultation
Your email address will not be published. Required fields are marked *


Content
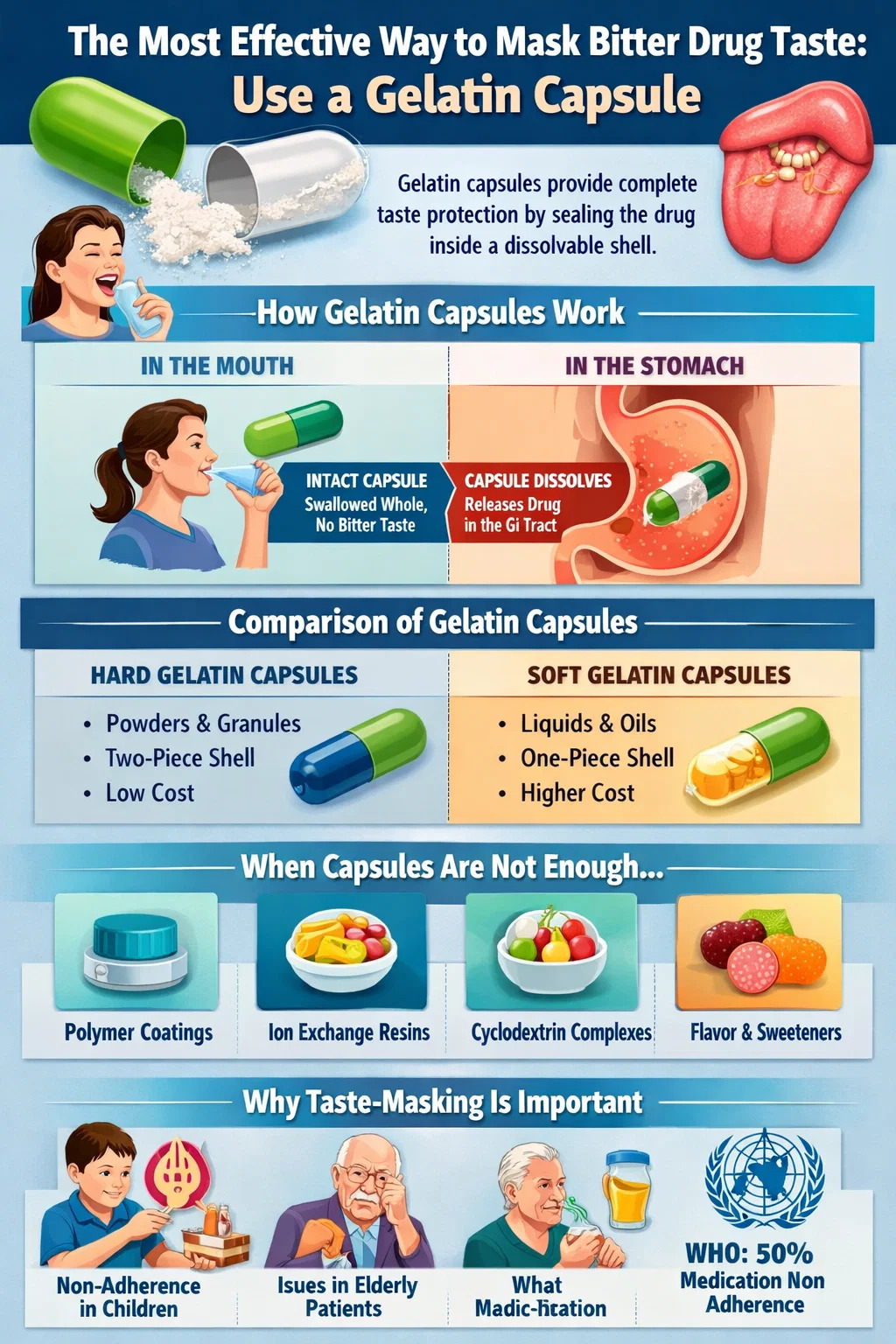
If you are dealing with a bitter-tasting medication — whether you are a pharmacist, a formulator, a caregiver, or simply a patient trying to swallow a pill without grimacing — the single most practical and immediately accessible solution is encapsulation in a gelatin capsule. By sealing the drug inside a hard or soft gelatin shell, the active ingredient never contacts the taste receptors on the tongue. The result is a near-complete elimination of perceived bitterness at the point of ingestion.
This approach works because bitterness is detected only when dissolved molecules interact with bitter-taste receptors (TAS2Rs) on the tongue. A sealed gelatin capsule bypasses that interaction entirely. The capsule dissolves downstream in the gastrointestinal tract, releasing the drug where absorption — not taste — is what matters.
Beyond gelatin capsules, a range of complementary and alternative strategies exist, including polymer coatings, ion exchange resins, cyclodextrin complexation, and flavoring systems. Each has its place depending on the dosage form, the patient population, and the severity of the bitterness problem. This article covers all of them in depth, starting with the most actionable.
Bitterness is not merely a sensory inconvenience. It is a measurable driver of medication non-adherence. Studies published in peer-reviewed pharmacy journals consistently show that taste aversion — particularly bitterness — is one of the top reasons patients, especially children and the elderly, refuse or discontinue oral medication regimens. According to the World Health Organization, non-adherence to chronic disease medication affects approximately 50% of patients, and palatability is a documented contributing factor.
In pediatric medicine, the problem is especially acute. Children have a higher density of taste buds and a lower threshold for bitterness than adults. Liquid formulations for children cannot easily rely on capsule encapsulation, which pushes formulators toward other taste-masking strategies. However, for patients who can swallow capsules — generally children older than five or six, adolescents, and adults — a gelatin capsule remains the most reliable first-line solution.
Bitter compounds include some of the most commonly prescribed drug classes: antibiotics such as clarithromycin and erythromycin, antiretrovirals, antihistamines like cetirizine, and many central nervous system drugs. The chemical basis of their bitterness varies — some are alkaloids, some are peptides, some are synthetic small molecules — but the problem they create in terms of patient experience is uniform.
A gelatin capsule is a shell made primarily from hydrolyzed collagen derived from animal connective tissue — typically bovine or porcine sources. Hard gelatin capsules consist of two interlocking cylindrical pieces (a body and a cap) that are filled with powder, pellets, granules, or small tablets. Soft gelatin capsules (softgels) are a single sealed unit typically filled with liquid or semi-solid material.
The gelatin shell is insoluble at room temperature but dissolves rapidly at body temperature in the presence of moisture. When a patient swallows a gelatin capsule with water, the shell remains intact in the mouth and esophagus — provided the patient does not chew it — and begins dissolving only once it reaches the warm, aqueous environment of the stomach. Dissolution typically occurs within 5 to 10 minutes of reaching the stomach, ensuring prompt drug release while providing complete taste protection during oral transit.
Hard gelatin capsules are the more versatile format. They accept a wide range of fill materials and are available in sizes ranging from 000 (the largest, holding approximately 1,400 mg) to 5 (the smallest, holding approximately 130 mg). Soft gelatin capsules are better suited for liquid drugs, oils, or compounds that benefit from dissolved presentation for faster absorption.
| Feature | Hard Gelatin Capsule | Soft Gelatin Capsule |
|---|---|---|
| Fill type | Powder, pellets, granules, mini-tablets | Liquids, oils, semi-solids |
| Shell construction | Two-piece (body + cap) | One-piece sealed unit |
| Taste masking effectiveness | Excellent (when sealed properly) | Excellent (hermetically sealed) |
| Suitable for moisture-sensitive drugs | Yes, with desiccant | Limited |
| Manufacturing complexity | Low to moderate | Moderate to high |
| Cost | Lower | Higher |
Selecting the correct capsule size is critical to ensuring the shell closes properly and maintains taste integrity. Overfilled capsules may not lock, allowing powder to leak out. Underfilled capsules leave air space that can cause the drug to shift and potentially cause the cap to disengage. Here is a reference table for standard hard gelatin capsule sizes:
| Capsule Size | Approximate Volume (mL) | Approximate Fill Weight (mg, typical powder) |
|---|---|---|
| 000 | 1.37 | Up to ~1,400 |
| 00 | 0.91 | Up to ~900 |
| 0 | 0.68 | Up to ~680 |
| 1 | 0.50 | Up to ~500 |
| 2 | 0.37 | Up to ~370 |
| 3 | 0.27 | Up to ~270 |
| 4 | 0.21 | Up to ~210 |
| 5 | 0.13 | Up to ~130 |
There are situations where a gelatin capsule alone does not fully resolve the bitterness problem. A drug with extremely high bitterness intensity — measured on the Beidler taste scale or assessed via electronic tongue instruments — may still produce taste aversion if trace amounts of drug vapor escape, if the capsule is opened for administration, or if the patient is unable to swallow solid oral dosage forms. In these scenarios, secondary or alternative taste-masking strategies become necessary.
One of the most robust pharmaceutical strategies is to coat individual drug particles or granules with a taste-masking polymer before filling them into a gelatin capsule. This double-barrier approach ensures taste protection even if the capsule is opened — for example, to mix the contents into food or a drink for patients who cannot swallow capsules.
Commonly used taste-masking polymers include:
Ion exchange resins bind ionizable drug molecules through electrostatic interactions, forming a drug-resin complex that releases the drug only under high ionic strength conditions — such as those found in gastrointestinal fluids — not in the low-ionic-strength environment of saliva. The most commonly used resin is Amberlite IRP69 (sodium polystyrene sulfonate), which is a strong cation exchanger suitable for basic (cationic) drugs.
This approach has been successfully applied to several well-known bitter drugs. Dextromethorphan, a bitter antitussive, is available in resin-complexed form in various over-the-counter cough preparations. The drug-resin complex can be suspended in liquid vehicles, making it suitable for pediatric liquid formulations — a context where capsule-based taste masking is often not applicable.
Cyclodextrins are cyclic oligosaccharides with a hydrophilic outer surface and a hydrophobic inner cavity. They can encapsulate lipophilic drug molecules within this cavity, forming an inclusion complex that reduces the free drug concentration in saliva and thereby reduces perceived bitterness. Beta-cyclodextrin and hydroxypropyl-beta-cyclodextrin (HP-β-CD) are the most widely used forms in pharmaceutical taste masking.
Cyclodextrin complexation also offers secondary benefits: improved drug solubility, enhanced chemical stability, and reduced irritation of the gastrointestinal mucosa. The complexes can be filled directly into gelatin capsules, combining the barrier effect of the capsule shell with the molecular encapsulation provided by the cyclodextrin — a particularly effective combination for highly bitter drugs.
Lipid-based systems — including solid lipid nanoparticles, lipid microparticles, and self-emulsifying drug delivery systems (SEDDS) — embed the drug within a lipid matrix that limits drug release in the oral cavity. These systems are often loaded into soft gelatin capsules, creating a dual taste-masking effect. The lipid matrix slows diffusion of drug molecules to saliva, while the gelatin shell provides an additional outer barrier.
Hot-melt extrusion (HME) is a solvent-free manufacturing process in which a drug is mixed with thermoplastic polymers and processed at elevated temperatures to form a solid dispersion. The resulting extrudate can be milled into granules and filled into gelatin capsules. HME-processed materials often exhibit reduced drug crystallinity, which changes solubility behavior and can reduce the rate of drug dissolution in the oral cavity — contributing indirectly to taste masking.
Flavoring and sweetening are not alternatives to physical taste masking — they are complements. When used inside gelatin capsule formulations (for example, in the powder fill blend), they can improve the olfactory experience even if the drug itself is not tasted. When used in liquid formulations where encapsulation is not feasible, they become a primary strategy.
Sweetness suppresses bitterness perception through a cross-modal sensory interaction. The most commonly used sweeteners in pharmaceutical formulations include:
Specific flavors are known to counteract bitterness more effectively than others. Vanilla, chocolate, strawberry, and cherry are the most frequently used pharmaceutical flavors, but their efficacy is drug-specific. Chocolate flavor is particularly effective against alkaloid-type bitterness because chocolate naturally contains bitter compounds (theobromine) and trained sensory responses already associate its flavor with accepted bitterness.
Cooling agents such as menthol create a sensory distraction that reduces bitterness perception by occupying the same trigeminal nerve pathways involved in taste processing. Menthol is used in various antitussive formulations precisely because it masks the bitterness of active ingredients like guaifenesin while also contributing to the therapeutic perception of the product.
A category of compounds known as bitter blockers or bitter taste receptor antagonists has emerged from research into the molecular biology of taste. These compounds bind to TAS2R receptors without activating them, competitively blocking the bitter stimulus. Adenosine 5'-monophosphate (AMP) and sodium gluconate are among the compounds shown to reduce bitterness perception in pharmaceutical contexts. GIV3727, a synthetic TAS2R antagonist, has been studied for blocking the bitterness of compounds like acesulfame K, saccharin, and certain drug molecules.
While bitter blockers are not yet a mainstream pharmaceutical excipient class, they represent a mechanistically precise approach that could become increasingly important as the taste receptor biology of specific drug molecules is better characterized.
For patients or formulators who prefer or require a non-animal-derived shell — for vegetarian, vegan, kosher, or halal compliance reasons — hydroxypropyl methylcellulose (HPMC) capsules, also called vegetable capsules or veggie caps, offer a functionally equivalent alternative to gelatin capsules for taste masking.
HPMC capsules are made from plant-derived cellulose and do not contain gelatin, starch, or preservatives. They are available in sizes equivalent to hard gelatin capsules (000 through 5) and perform comparably in terms of disintegration time and dissolution profile under standard conditions. One key difference is moisture behavior: HPMC capsules are less hygroscopic than gelatin capsules and maintain better dimensional stability under low-humidity storage conditions, making them preferred for moisture-sensitive drug substances.
Pullulan capsules — derived from the polysaccharide produced by the fungus Aureobasidium pullulans — are another plant-based option with a very low oxygen permeability, making them suitable for drugs that are sensitive to oxidative degradation. Their taste-masking performance is equivalent to gelatin for practical purposes.
Not all bitter drug problems occur in a pharmaceutical manufacturing context. Many patients encounter bitter taste when they are prescribed a drug in tablet or loose powder form and need a practical home solution. Some strategies are safe and effective; others carry risks that are worth knowing about.
Some patients are prescribed drugs in tablet form but have difficulty swallowing them. Crushing a tablet and mixing it with food is a common workaround, but it eliminates any taste protection the original coating provided. A better solution — where the drug is appropriate for compounding — is to have a compounding pharmacist repackage the drug dose into a gelatin capsule or an HPMC capsule. This is a legal, commonly performed pharmacy service and produces a product that completely hides the drug's taste during swallowing.
Important note: Not all tablets should be crushed or opened. Modified-release tablets, enteric-coated tablets, and certain cytotoxic or hormonal drugs must not be crushed. Always confirm with a pharmacist before attempting to repackage a drug.
Taste receptor sensitivity decreases at lower temperatures. Chilling a liquid medication — or swallowing a capsule with cold water — can reduce the intensity of bitter taste perception. This is especially useful for liquid pediatric formulations. Some parents administer liquid antibiotics directly from the refrigerator for this reason, though this should be confirmed as compatible with the specific drug's storage requirements.
When a drug can safely be taken with food, certain foods are more effective than others at masking bitterness. High-fat foods coat the oral mucosa, slowing drug dissolution in saliva. Acidic foods like orange juice can shift the ionization state of some basic drugs, reducing their interaction with bitter taste receptors. Chocolate spread, honey, and fruit purées are commonly used vehicles for pediatric liquid drugs.
However, drug-food interactions are a real concern. Grapefruit juice, dairy products, and high-calcium foods interact with specific drugs (fluoroquinolone antibiotics, tetracyclines, and others) in ways that significantly reduce absorption. The food vehicle should always be confirmed as safe with the specific medication.
Flavor perception is approximately 80% olfaction. Pinching the nose while swallowing a liquid medication dramatically reduces perceived flavor, including bitterness components that arise from retronasal olfaction. This is a simple, zero-cost strategy that is effective for adults and older children who can follow the instruction.
Another approach is to apply a small amount of a numbing agent to the tongue prior to drug administration. OTC topical anesthetics containing benzocaine or lidocaine can temporarily reduce taste sensitivity. This is occasionally used in clinical settings for patients receiving very bitter oral chemotherapy drugs, though it is not commonly recommended for routine home use.
Effective taste masking starts with measuring the bitterness of the drug substance. Several methodologies are available, ranging from human sensory panels to electronic instruments.
Trained human taste panels remain the gold standard for evaluating bitterness intensity and taste-masking efficacy. Panelists assess samples against a reference scale — often using caffeine or quinine hydrochloride solutions as reference bitterness standards. Quinine hydrochloride is detectable by most humans at concentrations as low as 0.008% w/v, making it a useful benchmark for highly bitter drugs.
The obvious limitation of human panels is safety — early-stage drug candidates may not yet have full safety profiles, making human oral exposure inadvisable. This pushes formulators toward instrumental alternatives in early development.
Electronic tongues (e-tongues) use arrays of cross-reactive chemical sensors — typically lipid-polymer membranes or electrochemical electrodes — to generate a signal fingerprint that correlates with taste characteristics including bitterness. Systems such as the Insent TS-5000Z (developed in Japan) use sensor arrays specifically calibrated for human taste receptor responses and are capable of measuring bitterness intensity in pharmaceutical preparations without human subjects.
E-tongue measurements can be performed on formulated drug products — including capsule contents dissolved in simulated saliva — allowing formulators to compare the bitterness of masked versus unmasked preparations quantitatively. This data supports regulatory submissions and quality control testing.
Computational models that predict bitterness from molecular structure are increasingly used in early drug discovery. Databases such as BitterDB catalog bitter compounds and their structural features, allowing machine learning models to flag new chemical entities likely to be bitter before synthesis. This type of upstream awareness helps formulators plan taste-masking strategies — including gelatin capsule selection — earlier in the development process, reducing the cost of reformulation later.
Every excipient used in a taste-masking strategy — whether it is a gelatin capsule shell, a polymer coating, a sweetener, or a flavor — must meet regulatory requirements for safety, purity, and function. The primary frameworks governing pharmaceutical excipients include:
Gelatin sourced from bovine or porcine materials is subject to transmissible spongiform encephalopathy (TSE) and BSE (bovine spongiform encephalopathy) risk assessments under EMA guidelines. Manufacturers of gelatin capsules are required to document the geographic origin and processing history of raw materials. This regulatory burden is one reason some manufacturers have migrated to HPMC capsules, which carry no TSE concerns.
For pediatric formulations specifically, the EMA Pediatric Committee (PDCO) and the FDA's Office of Pediatric Therapeutics have issued guidance recommending that taste and palatability be formally assessed and documented during the development of oral pediatric drug products. This has elevated taste masking from a formulation convenience to a regulatory expectation in the pediatric drug development context.
With multiple strategies available, selecting the right approach requires considering the drug's physicochemical properties, the target patient population, the intended dosage form, and the manufacturing resources available. The following framework provides practical guidance:
The cost and complexity of taste-masking strategies also varies significantly. Hard gelatin capsule filling is among the cheapest and simplest pharmaceutical manufacturing operations. Hot-melt extrusion with polymer coating followed by capsule filling is substantially more complex and requires specialized equipment. Choosing the simplest strategy that achieves acceptable taste masking is not just economically sensible — it also reduces the number of variables in the formulation, simplifying regulatory submission and quality control.
Your email address will not be published. Required fields are marked *
If you would like to learn more about our products, please feel free to contact us and we will do our to assist you.
No.1 Tianzhu 3rd Road, Dufu Town, Xinchang County, Zhejiang Province
86-575 8606 0065
86-159 8825 2009
+86 159 8825 2009
+1 380 215 7432
