Product Consultation
Your email address will not be published. Required fields are marked *


Content
Capsules — particularly gelatin capsules — are manufactured through a dip-molding process in which precision-engineered stainless steel pins are submerged into a temperature-controlled gelatin solution, withdrawn, dried, stripped, trimmed, and joined to form the finished shell. This process applies to hard gelatin capsules (HGC), which account for roughly 70–75% of all capsules produced globally. Soft gelatin capsules (softgels) follow a different rotary die encapsulation method where the shell and fill material are formed simultaneously.
The entire manufacturing cycle for hard gelatin capsules — from pin dipping to finished shell — typically takes 45 to 50 minutes per batch cycle on a modern automated line, with a single production machine capable of producing over 1 million capsule shells per hour. Understanding this process is essential for pharmaceutical manufacturers, nutraceutical brands, compounding pharmacies, and anyone sourcing or evaluating capsule materials.
A gelatin capsule is a shell made primarily from gelatin — a protein derived from the partial hydrolysis of collagen sourced from animal bones, skins, and connective tissues, most commonly from bovine or porcine origins. The shell dissolves in gastric fluids within 3 to 10 minutes, releasing its contents efficiently into the digestive system.
Gelatin capsules dominate the pharmaceutical and supplement market for several measurable reasons:
Two primary types of gelatin capsules exist in commercial production: hard gelatin capsules and soft gelatin capsules. Their manufacturing processes are fundamentally different, and their applications reflect those differences.
Before examining the production process, it is necessary to understand the input materials that directly determine shell quality, dissolution performance, and regulatory compliance.
Pharmaceutical-grade gelatin used in capsule production must meet the specifications outlined in the United States Pharmacopeia (USP), European Pharmacopoeia (Ph. Eur.), and Japanese Pharmacopoeia (JP). Key quality parameters include:
Pure gelatin shells would be too brittle for practical handling. Plasticizers are added to introduce flexibility. For hard capsules, plasticizer content is kept minimal (below 1–2%). For soft gelatin capsules, plasticizer concentration is much higher — typically 20–30% of the shell formulation. The most common plasticizer is glycerin, with sorbitol used in humidity-sensitive applications.
Titanium dioxide is used as an opacifier to prevent light degradation of photosensitive fill materials. Approved colorants (FD&C dyes, iron oxides, natural pigments such as annatto or carmine) are incorporated into the gelatin solution before dipping. A single capsule shell may contain 0.01% to 2% w/w titanium dioxide depending on the required opacity level.
Purified water (meeting USP or Ph. Eur. standards) is the primary solvent for gelatin dissolution. The gelatin solution used in dipping typically contains 30–40% w/w gelatin solids dissolved in water, maintained at 50–60°C to keep the solution at appropriate viscosity for dipping.
Some manufacturers add low-concentration antimicrobial preservatives such as sulfur dioxide (SO₂) to the gelatin mass to prevent microbial contamination during processing. The permitted level in finished gelatin is typically not more than 50 mg/kg per Ph. Eur. specifications.
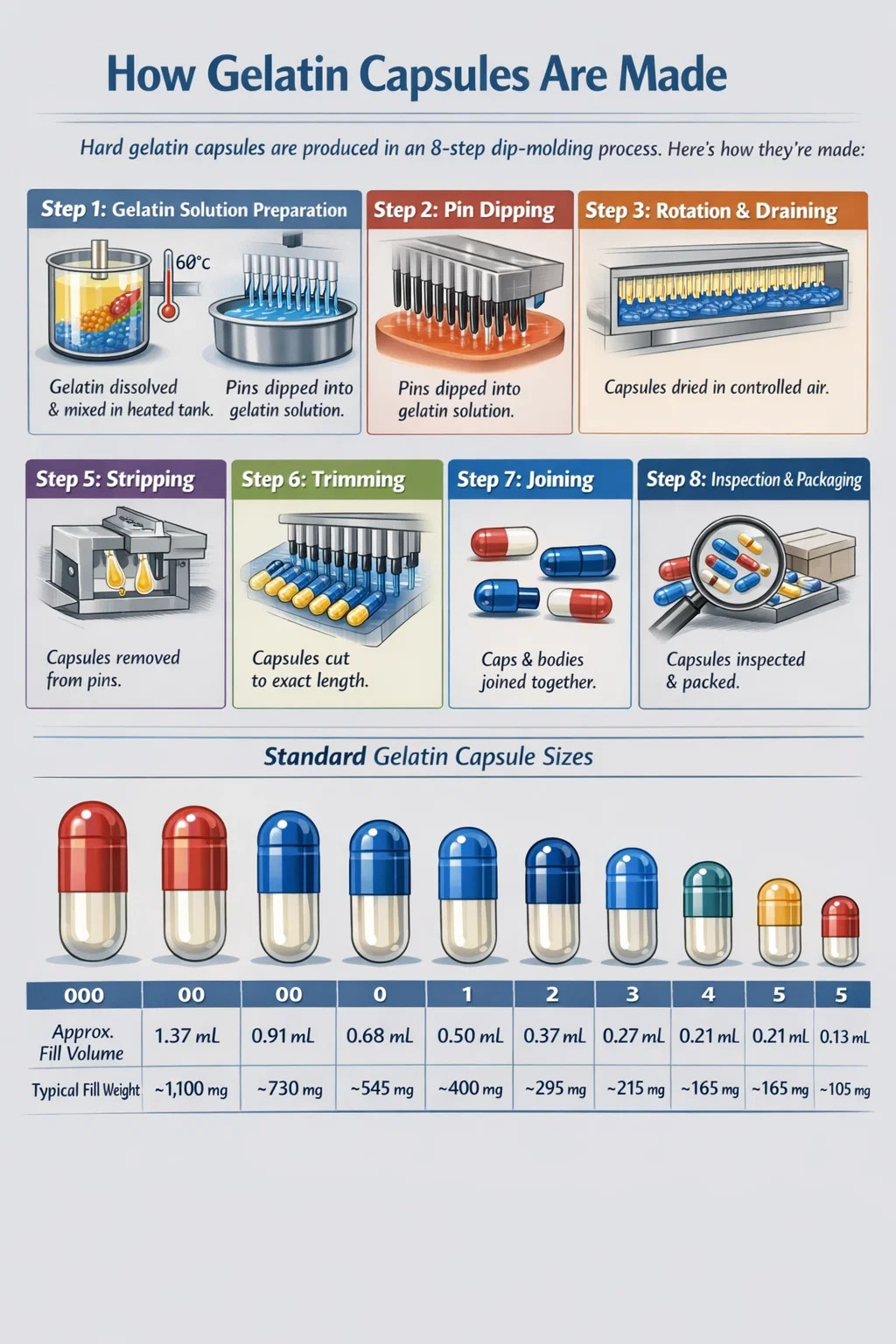
The manufacturing of hard gelatin capsule shells follows a tightly controlled sequence of steps. Each phase occurs within a dedicated section of an automated capsule-making machine (such as those produced by Capsugel, ACG, or Qualicaps). Here is a detailed breakdown of each stage:
Pharmaceutical-grade gelatin granules are weighed and dissolved in purified water using a jacketed mixing vessel with controlled heating. The solution is brought to 60–70°C under continuous stirring for 2–4 hours until fully homogeneous. Colorants, opacifiers, and any excipients are added during this stage. The final solution is degassed under vacuum to remove air bubbles that would otherwise create pinholes in the finished shell. The solution is then transferred to a holding tank where temperature is maintained at 50–55°C to prevent premature gelling while maintaining correct viscosity for dipping.
The core of hard capsule manufacturing is the dip-molding process. Stainless steel or brass pins — precision-machined to the exact dimensions of each capsule size (size 000 to size 5, with corresponding volumes from 1.37 mL down to 0.13 mL) — are arranged in bars holding hundreds of pins each. These pin bars are first cleaned, inspected, and pre-lubricated with a release agent (commonly a mold lubricant based on cetrimide or similar agents) to facilitate capsule removal.
The pin bars are then dipped into the temperature-controlled gelatin solution. The dipping tank is maintained at 45–50°C to ensure the gelatin coats the pins uniformly. Body pins and cap pins are dipped separately, as they have slightly different dimensions — the cap has a marginally larger diameter to allow the body to slide inside and lock during joining. Dipping depth and withdrawal speed are precisely controlled to achieve a uniform shell wall thickness of 0.09 to 0.12 mm.
Immediately after dipping, the pin bars are inverted and rotated slowly. This rotation ensures even distribution of the gelatin film over the entire pin surface, preventing pooling at the tip. During this phase, excess gelatin drains back into the dipping tank, reducing material waste.
The coated pins pass through a series of drying kilns where conditioned air at precisely controlled temperature and humidity is directed over the gelatin film. Drying conditions are typically 20–25°C with relative humidity maintained between 30–45%. Too-rapid drying causes cracking; insufficient drying leaves the shells soft and sticky. The drying tunnel on a modern automated machine may be 30 to 50 meters in effective length, with multiple temperature and humidity zones, to achieve gradual, uniform drying. This stage typically takes 30–40 minutes of total tunnel residence time.
Once dried to the correct moisture content (typically 13–16% w/w at this stage), the semi-rigid gelatin shells are mechanically stripped from the pins using rubber stripping jaws that grip and pull the shell off without distortion. This is a critical step — any pin surface irregularities or inadequate lubrication will cause tearing or distortion at this point.
The stripped shells have an uneven open end (the end that was at the tip of the pin). Precision rotary knives trim each shell to the exact specified length. Hard gelatin capsule cap and body lengths are standardly defined — for example, a size 0 capsule has a body length of 18.0 mm and a cap length of 11.7 mm (approximate standard dimensions; exact specs vary by manufacturer). Trimming accuracy is typically within ±0.3 mm.
Trimmed bodies and caps are fed into a joining section where they are pre-locked: the body slides into the cap to a pre-lock position, keeping them together as empty shells during handling and filling operations. Pre-locked capsules can be separated easily by filling machines for filling, then snapped fully closed after the fill is complete. The joining mechanism uses guides and cam followers to push the body into the cap at a controlled depth.
Finished empty gelatin capsule shells pass through automated inspection systems equipped with cameras and sensors that detect visible defects — pinholes, double walls, splits, dents, and color inconsistencies. Rejection rates on well-maintained modern lines are typically below 0.5%. Accepted shells are bulk-packed in polybags within cardboard cartons and stored at controlled conditions of 15–25°C and 35–65% RH to maintain shell integrity during shelf life (typically 3–5 years).
Hard gelatin capsules are manufactured in standardized sizes, each corresponding to a defined fill volume capacity. Choosing the correct size is a critical formulation decision.
| Capsule Size | Approximate Fill Volume (mL) | Typical Fill Weight (mg) at 0.8 g/mL density | Common Applications |
|---|---|---|---|
| 000 | 1.37 | ~1,100 | High-dose veterinary, nutraceuticals |
| 00 | 0.91 | ~730 | Fish oil, herbal extracts |
| 0 | 0.68 | ~545 | Most common OTC supplement size |
| 1 | 0.50 | ~400 | Pharmaceutical Rx products |
| 2 | 0.37 | ~295 | Mid-dose formulations |
| 3 | 0.27 | ~215 | Potent APIs, pediatric formulations |
| 4 | 0.21 | ~165 | Very high potency drugs |
| 5 | 0.13 | ~105 | Neonatal and micro-dosing |
Soft gelatin capsules (softgels) are produced using the rotary die encapsulation process, invented by Robert Pauli Scherer in 1933. Unlike hard capsule manufacturing, the softgel process forms the shell and encapsulates the fill material simultaneously in a continuous operation.
A warm gelatin mass (containing gelatin, plasticizer — typically glycerin and/or sorbitol at 20–30% of dry gelatin weight — and water) is spread onto cooled casting drums to form continuous gelatin ribbons of controlled thickness, typically 0.5 to 1.2 mm thick. Two ribbons are formed simultaneously — one for each half of the capsule.
The two gelatin ribbons are fed from opposite sides into a rotary die mechanism consisting of two counter-rotating die rolls. As the ribbons converge at the dies, fill material (liquid, suspension, or paste) is injected from a fill pump through a wedge-shaped injection mechanism positioned between the ribbons. The dies stamp and seal the gelatin around the fill material using heat and pressure — typically 37–40°C at the die surface — while simultaneously cutting the capsule from the ribbon. This produces a hermetically sealed capsule in a single operation.
Freshly formed softgels contain 30–40% moisture and must be dried. They are transferred to tumble dryers — large rotating drums with conditioned air flow — where they tumble continuously for 2–4 hours to prevent sticking and promote uniform drying. Subsequent tray drying may continue for 24–48 hours in controlled environments until the shell reaches equilibrium moisture content of approximately 6–10% w/w.
The fill for a soft gelatin capsule must be liquid or semi-solid at processing temperatures. Materials that are incompatible with softgel filling include:
Hydroxypropyl methylcellulose (HPMC) capsules — also called vegetarian or vegan capsules — have gained significant market share as alternatives to gelatin capsules, particularly for products targeting vegetarian, vegan, halal, and kosher consumers. The manufacturing process for HPMC capsules uses the same dip-molding principle, but with important differences.
| Parameter | Gelatin Capsule | HPMC Capsule |
|---|---|---|
| Shell material | Animal-derived gelatin | Plant-derived cellulose polymer |
| Dissolution time (gastric) | 3–10 minutes | 15–30 minutes |
| Moisture sensitivity | High (brittle below 12% RH, soft above 65% RH) | Lower — more stable across RH range |
| Cost per unit | $0.01–$0.05 | $0.03–$0.10 (approx. 2–3× higher) |
| Filling speed compatibility | Excellent — runs on all standard fillers | Good — may require speed adjustments |
| Cross-linking risk | Yes — aldehydes and reducing sugars can cross-link shell | No — not susceptible to cross-linking |
| Dietary suitability | Not suitable for vegans/vegetarians | Suitable for all dietary preferences |
One of the most practically significant challenges in gelatin capsule manufacturing and formulation is shell cross-linking — a chemical reaction where aldehyde-containing compounds (from excipients, fill materials, or packaging) react with the amino groups in the gelatin protein chains, forming covalent bonds that make the shell rigid and resistant to dissolution.
Cross-linked gelatin shells may pass initial dissolution testing but fail after 3–6 months of storage, creating a serious bioavailability risk that is often not detected until stability studies are well underway. The USP two-tier dissolution test was specifically developed to address this: capsules that fail standard dissolution criteria are tested again in the presence of enzymes (pepsin or pancreatin) to determine if cross-linking is the cause.
Known triggers for cross-linking in gelatin capsule systems include:
Mitigation strategies include replacing lactose with non-reducing sugars (e.g., mannitol or microcrystalline cellulose), using desiccants and inert headspace gases in packaging, selecting HPMC capsules for aldehyde-susceptible formulations, and conducting accelerated stability testing at 40°C/75% RH for 6 months as a standard protocol.
Capsule manufacturers supplying the pharmaceutical industry must operate under cGMP (current Good Manufacturing Practice) regulations as defined by FDA 21 CFR Parts 210/211 (US), EudraLex Volume 4 (EU), or equivalent national guidelines. The quality control regime for gelatin capsule production covers several dimensions:
Beyond standard hard and soft gelatin capsules, the capsule industry has developed specialized variants to meet specific drug delivery requirements. These modifications are introduced either during shell manufacturing or as post-production treatments.
Standard gelatin capsules dissolve rapidly in gastric acid (pH 1–2). For drugs that are acid-labile or must be delivered to the intestine, gelatin capsules are coated post-filling with enteric polymers such as hydroxypropyl methylcellulose phthalate (HPMCP), cellulose acetate phthalate (CAP), or Eudragit L100. These coatings remain intact at gastric pH below 5.0 but dissolve at intestinal pH above 5.5–6.0, enabling targeted intestinal release.
Hard gelatin capsules can be filled with liquid or semi-solid formulations — a technology that bridges the simplicity of hard capsule manufacturing with the bioavailability advantages traditionally associated with softgels. Liquid fills for hard capsules must be non-aqueous or contain less than 10% water to prevent shell softening. After filling, capsules are sealed using banding (a gelatin band applied around the cap-body junction), heat sealing, or spin welding. This format has shown particular utility for improving the bioavailability of BCS Class II and IV poorly soluble drugs through lipid-based drug delivery systems (LBDDS).
Beyond HPMC, alternative shell materials are available for specific applications. Pullulan capsules (made from fermented tapioca) offer excellent oxygen barrier properties — oxygen transmission rates approximately 10 times lower than gelatin — making them suitable for highly oxidation-sensitive fill materials. Starch-based capsules are another animal-free alternative but are less commonly used due to higher brittleness and processing challenges.
The gelatin capsule shell itself is not typically used to achieve modified release — that function is usually built into the fill (coated pellets, mini-tablets, controlled-release granules). However, the gelatin capsule is an ideal container for housing multiple pellet populations with different release profiles. For example, a size 0 hard gelatin capsule can contain a blend of immediate-release and 8-hour extended-release pellets in a single capsule, creating a biphasic release profile from a simple manufacturing step.
The empty gelatin capsule shell is only part of the story. The filling process — carried out at pharmaceutical manufacturers, contract development and manufacturing organizations (CDMOs), or compounding pharmacies — is equally critical to the final product quality.
Industrial capsule fillers (from manufacturers such as IMA, Bosch/Syntegon, and MG America) operate on a continuous or intermittent motion basis. They open the pre-locked capsule shells, fill the body with the dose of powder, pellets, or liquid, and then close and eject the filled capsule. Output rates for high-speed rotary fillers range from 150,000 to 500,000 capsules per hour. Weight variation control is achieved through automated checkweighers that reject capsules outside the specification tolerance, typically ±5% of target fill weight for powder fills.
For small-batch compounding or clinical trial manufacturing, capsule filling plates (Torpac, Capsule Machine) allow manual filling of 50 to 300 capsules per batch. Semi-automatic bench-top fillers bridge the gap, handling 5,000 to 50,000 capsules per hour and are common in small-scale pharmaceutical operations and specialty compounding settings.
Hard gelatin capsule filling must occur in controlled environments. The most critical parameter is relative humidity: gelatin shells become too brittle to handle below 30% RH and too soft and tacky above 60% RH. Standard filling room conditions are 20–25°C and 40–55% RH. In humid climates, filling rooms require active dehumidification with validated HVAC systems.
For pharmaceutical companies, nutraceutical brands, and CDMOs, selecting the right gelatin capsule supplier requires evaluation across several dimensions beyond basic price per thousand capsules.
Your email address will not be published. Required fields are marked *
If you would like to learn more about our products, please feel free to contact us and we will do our to assist you.
No.1 Tianzhu 3rd Road, Dufu Town, Xinchang County, Zhejiang Province
86-575 8606 0065
86-159 8825 2009
+86 159 8825 2009
+1 380 215 7432
